


Looking to Buy Tickets?
Easiest event ticketing platform around
Create a free account and manage your event from start to finish with our easy-to-use ticketing platform. With TicketLeap, you’ll have access to our comprehensive suite of event management features from the get-go. What's more, you can conveniently manage box office ticket sales and check-in attendees from our mobile app, giving you the freedom and flexibility to stay connected and in control, no matter where you are!
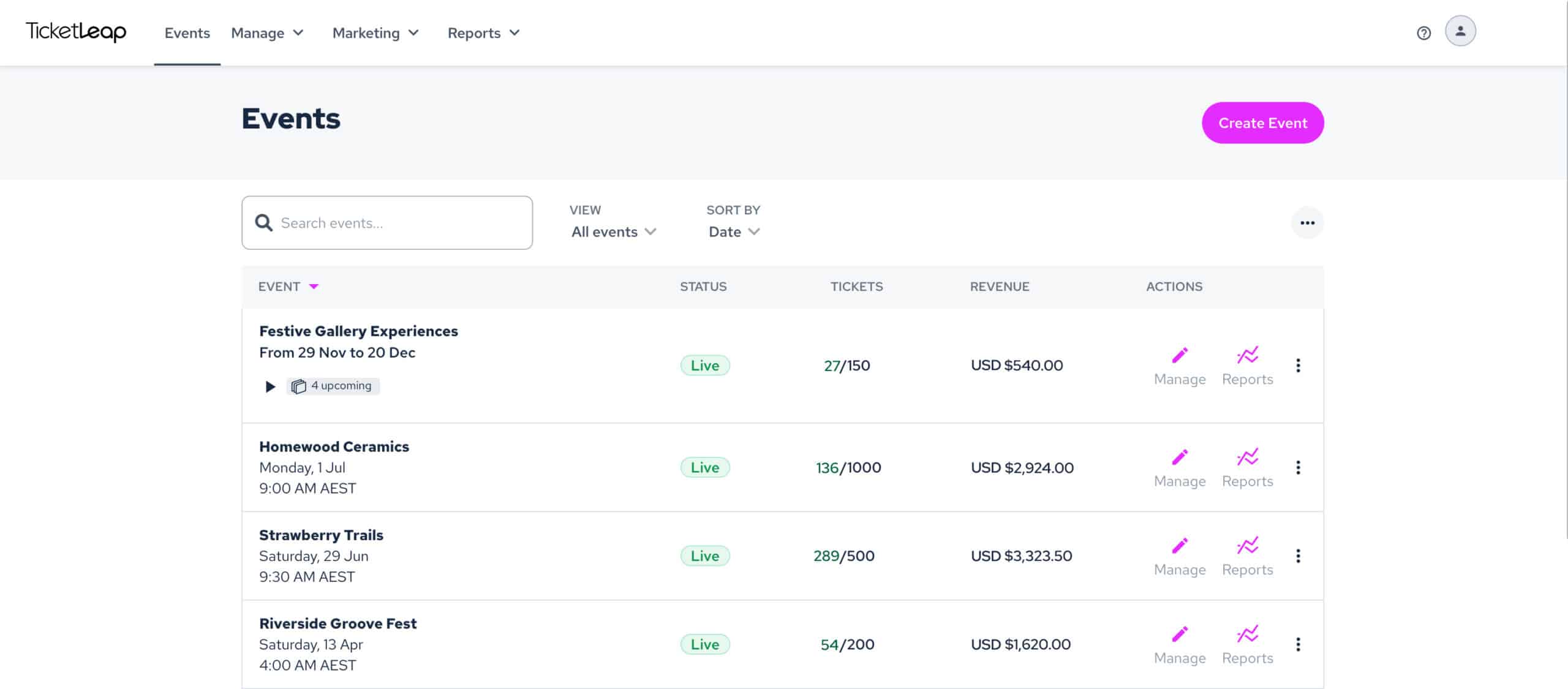
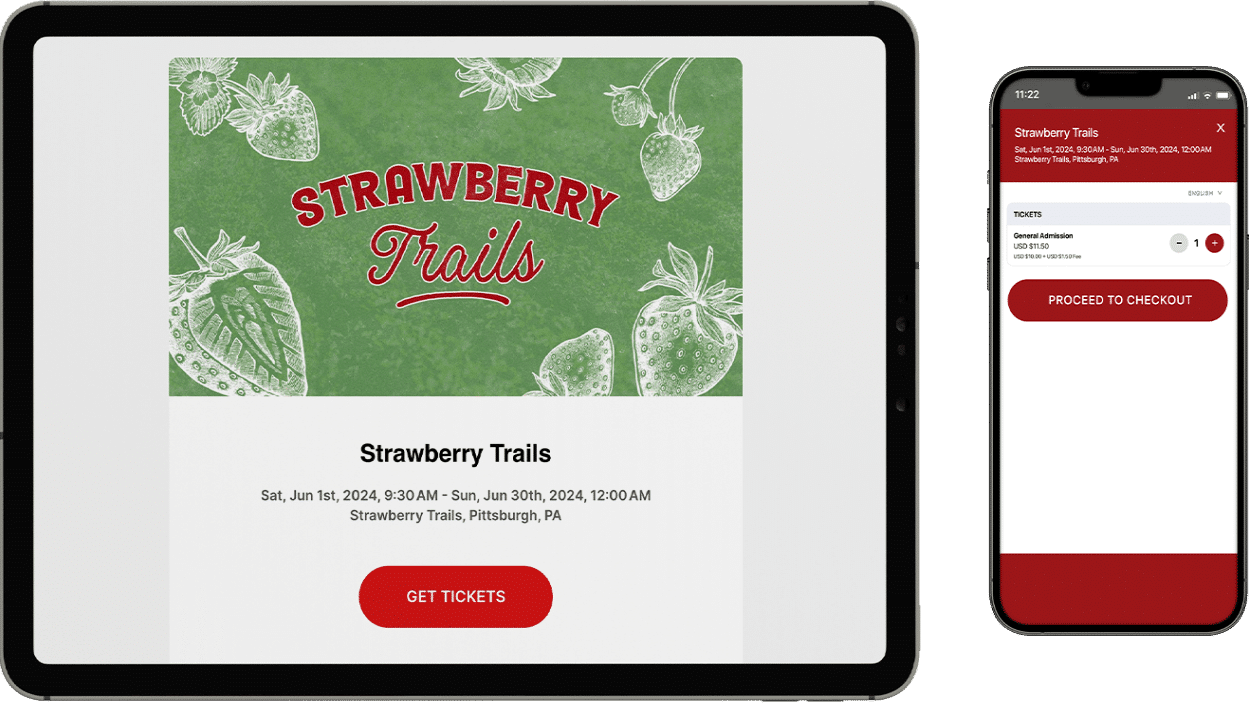

All the tools you need for online ticket sales
Say goodbye to restrictive service tiers and hello to a comprehensive set of free ticketing features! With no contracts, subscriptions, or commitments, you can effortlessly create beautiful event pages, efficiently manage your events, and start selling tickets online in just a matter of minutes.
Experience the convenience and flexibility you deserve with our hassle-free event ticketing platform. Don't miss out on this opportunity to streamline your event ticketing and maximize your event's success!



For over 20 years, we’ve helped event organizers sell more than 90 million tickets!






Resources for event creators
Unlock the magic behind seamless event planning with our blog! Packed with impactful event marketing tips, expert event management advice, and insights on emerging event trends, TicketLeap is the ultimate resource for mastering online ticket sales!



Comprehensive Help Center: Get the answers you need, fast!
Our user-friendly TicketLeap help center is always buzzing with the latest updates to empower you to plan your next event with confidence! It's also a handy resource to use to answer ticket buyer questions. Trust us to provide the unwavering support you need for a successful event!







